The Adsorber
 The CytoSorb adsorber is a CE-marked Class IIb medical device that is specifically approved for the removal of cytokines, bilirubin and myoglobin. Next to this CytoSorb is indicated for use intraoperatively during cardio-pulmonary bypass surgery for the removal of P2Y12-Inhibitor Ticagrelor and/or Factor Xa-Inhibitor.
It is approved for use with standard hemodialysis-, hemofiltration-, ECMO- and heart lung-machines found in hospitals worldwide.
CytoSorb comprises of several important features: An advanced biocompatible and hemocompatible porous polymer sorbent bead technology with extremely high binding capacity, documented ability to help to reduce a broad range of cytokines and other inflammatory mediators, bilirubin and myoglobin, and has good safety data in thousands of human treatments.
In regards to cytokine removal, the goal is to reduce cytokines to levels that are no longer toxic, while still keeping the immune system intact.
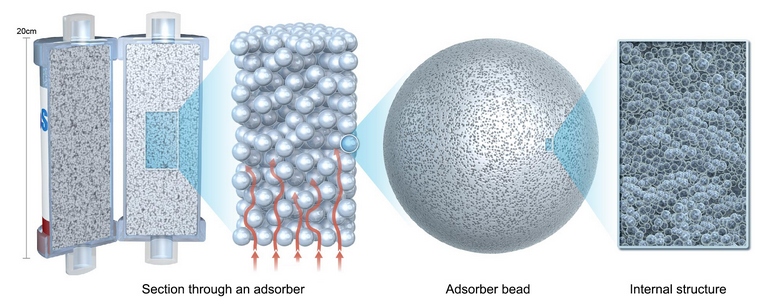
The content of the CytoSorb cartridge consists of a highly porous high-tech polymer. The tiny beads can bind to a wide range of inflammatory mediators such as e. g. cytokines, chemokines, and anaphylline toxins by virtue of their structure.
The CytoSorb adsorber is a CE-marked Class IIb medical device that is specifically approved for the removal of cytokines, bilirubin and myoglobin. Next to this CytoSorb is indicated for use intraoperatively during cardio-pulmonary bypass surgery for the removal of P2Y12-Inhibitor Ticagrelor and/or Factor Xa-Inhibitor.
It is approved for use with standard hemodialysis-, hemofiltration-, ECMO- and heart lung-machines found in hospitals worldwide.
CytoSorb comprises of several important features: An advanced biocompatible and hemocompatible porous polymer sorbent bead technology with extremely high binding capacity, documented ability to help to reduce a broad range of cytokines and other inflammatory mediators, bilirubin and myoglobin, and has good safety data in thousands of human treatments.
In regards to cytokine removal, the goal is to reduce cytokines to levels that are no longer toxic, while still keeping the immune system intact.
The content of the CytoSorb cartridge consists of a highly porous high-tech polymer. The tiny beads can bind to a wide range of inflammatory mediators such as e. g. cytokines, chemokines, and anaphylline toxins by virtue of their structure.
Functionality
In CytoSorb therapy*, the blood is purified by adsorption. With a total surface of > 45,000 square meters (> 4 large football fields), the adsorber is able to effectively and over time (up to 24 hours) lower high cytokine levels. It is mainly hydrophobic (water-insoluble) molecules that can be removed up to a size of approximately 60kDa. These include cytokines. The removal of these substances is concentration-dependent, which helps to prevent complete removal of physiologic mediators.
High Inflammatory mediators plasma levels are lowered to support immune modulation to regain a re-balanced situation.
In addition, other metabolic products such as bilirubin or myoglobin can be removed from the bloodstream. Particularly important substances, such as albumin, remain largely unaffected.
Setup
CytoSorb is a whole blood adsorber. The unique design of the CytoSorb cartridge and its adsorber beads from the basis for its functionality and high biocompatibility.
All for more information about setup and integration option go to “Setup”

